Hemodialysis eGFR Equivalence: Why More—and More Often—Matters

In a recent post, I compared the weekly volume of blood presented to two normal kidneys with the blood volume presented to a dialyser for a composite 3 times weekly dialysis. In highlighting this, I compared the exquisite evolutionary physiology of two healthy, no-fuss, subliminally functioning kidneys with our as yet far-from-perfect efforts to emulate the intricacies of nature: my code for Shakespeare’s “what a piece of work is a man.”
But, there is much more to native kidney function and to the mechanical models we have devised to attempt to mirror that function than simply to contrast the available volume of blood delivered for “processing” over any given time period. Stark as the differences in volume are, it is how that presented volume is then modified, processed, and converted to an output that truly determines the end efficiency of the “nature versus man” model.
The conversion of blood flow (volume) to clearance (cleansed volume) is a far more complex masterpiece again…and one we still fall far short of emulating. But, dazed by self-satisfaction in our seeming ingenuity, our very human tendency to over-promise outcomes can lead to significant consumer disappointment as our imperfect models under-deliver.
We must learn to better fess up to the users of all dialysis systems that dialysis will help—to a point—but it is still far from the real deal. Understanding and acknowledging this from the outset might place both provider and consumer on a more empathetic footing, each for the other. The treatment “giver” will better understand and can temper unrealistic expectations in the receiver, while the receiver can be more mindful of the limitations of the giver as that treatment is administered.
But, I digress. Let me get back to clearance.
First, what is a filter? In its simplest interpretation, a filter is a separator.
The air filter of a car is a separator: it traps the dust as it lets the clean air through.
A coffee filter is a separator: it traps the grounds while the water and the coffee flavor pass through.
A fly-wire screen is a separator: it keeps the flies and mozzies out, even as the air passes on through.
Filters get “stuffed up” in one of three ways. Take the fly-wire screen as our example:
Punch holes in the screen? The air still freely passes back and forth but now, some of the flies and mozzies will too. The filter leaks—as kidneys do when they leak stuff they shouldn't, like protein.
Paint the fly-wire with paint? Now nothing gets through—air, flies, or mozzies. The filter is blocked, as happens when the kidneys fail to remove the wastes they should, like in renal failure.
Do a bit of both? Then the filter will both leak and poorly filter—a classical case being diabetic nephropathy.
So it is with the human kidney: diseases that we neither expect nor want will cause the filter systems of the human kidney—the glomeruli—to succumb to either or both of these injuries.
Thus, even though the blood volume delivered to the kidneys may not change, the capacity to filter and modify it does change, progressively, as more and more damage is done by whatever disease process is underway. At the end of the day, while the disease process does matter, especially as it may or may not be variably treatable, it is the progressive loss of filter function that really counts.
So, what is that function, and what models can we use to demonstrate “function” as we seek to replace it? For now, forget about all the other magic stuff the kidneys do:
The release of renin and subsequent cascade of substances that modify blood pressure control
The manufacture of erythropoietin (EPO)
The conversion of vitamin D from its inactive to active form
The manufacture of vasodilatory prostaglandins
The list goes on, and each is important in its own right, but for now, let us think only about the control of waste and volume. And, as we think of waste and volume, let us think about how we have sought to artificially replace these functions by dialysis. Clearly, both matter. And, both—to an extent—depend on different factors.
First: Think Solutes
Waste(s), the by-product(s) of daily cell metabolism, must be excreted – removed. This is an imperative of kidney function.
Of these wastes, many are small and relatively easy-to-remove – like urea, potassium, and to a lesser extent but in the same group, creatinine. Of course, there are hundreds of others, but these will do as examples.
Other wastes – the so-called “middle molecules” – include beta-2 microglobulin (B2M) and phosphate, the latter of which is a small molecule that behaves more like a much larger one due to a firmly-adherent coating of water molecules that slow its filtration progress up.
Finally, there are complex, large wastes, many of them bound to protein, that filter very slowly indeed.
Small solute clearance is “modeled” by the example of urea. Urea “clearance” across a dialysis membrane can be determined mathematically by assessing the amount that appears in the dialysis fluid from a known quantity of urea in the blood as dialysis progresses. A complex mathematical concept and equation (we know it as Kt/Vurea) is used to estimate the amount of urea removed (K) in a given time (t) from a known volume of urea (V). Then, depending on the number of repetitive dialysis sessions in a week, a graph emerges that pictorially represents urea removal, per session, and per week relative to the number of sessions provided in that week.
Study the graph below. Accept—for now—that a Kt/Vurea of 1.3 is regarded as “adequate” clearance for any one session. Then, follow upwards the Kt/V line of 1.3, effectively the amount of urea removed (vs. renal clearance) for 1x, 2x, 3x, etc. sessions per week. This graph from the work of Casino and Lopez(1), which I have adapted for this blog by adding some colored annotations, should help to show you the cumulative effect of more and more sessions/week on urea removal for any given Kt/Vurea.
The cumulative removal of urea is—in effect—the renal urea clearance (the vertical axis on the left) and represents the effective GFR generated over a week by each equal treatment, simply by the application of more treatments.
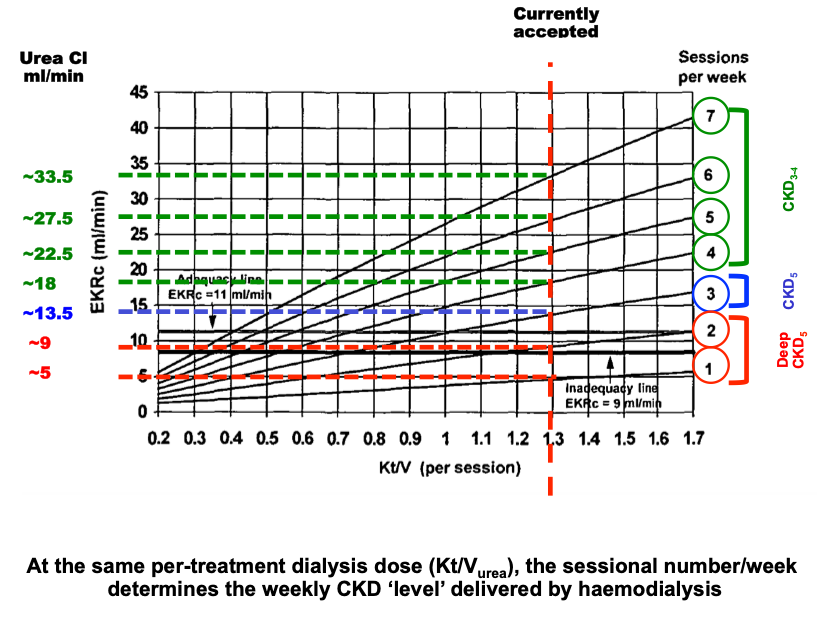
In essence, what this graph is saying is that if a Kt/Vurea of 1.3 (the “adequate” urea removal target for any one session) is achieved once a week, this will provide an equivalent clearance of ~5 ml/min (= deep CKD5).
With 2 sessions, this increases to ~9 ml/minute.
With 3 sessions, weekly equivalent clearance is ~13 ml/min = still well inside CKD5.
The more sessions per week, the greater the weekly urea clearance, such that with 7 sessions, a weekly GFR equivalent of ~33ml/min (i.e., borderline CKD3-CKD4) is achieved.
Now, while this is a stylized representation, it is nonetheless likely close to the truth. And, as it applies to small solute clearance, sessional number to a standardized clearance per session governs dialysis efficacy.
So, what the Casino and Lopez study means is that more is better.
What, then, of middle molecular clearance? Here, let me use phosphate as the example. Many years ago, Gutswiller et al(2) performed a careful study of phosphate clearance that still provides our model for phosphate removal. Oddly, though, phosphate behaves quite differently to urea when it traverses a dialyser membrane. Phosphate does pass moderately well, to begin with, but then its appearance time in the dialysis fluid quite rapidly slows, after an hour or so of steady dialysis, from a “quick whoosh” to a “slow ooze.” And from that point on, it just keeps oozing.
The following graph (adapted from Gutzwiller et al) shows this clear “switch” from quick mover to slow oozer.
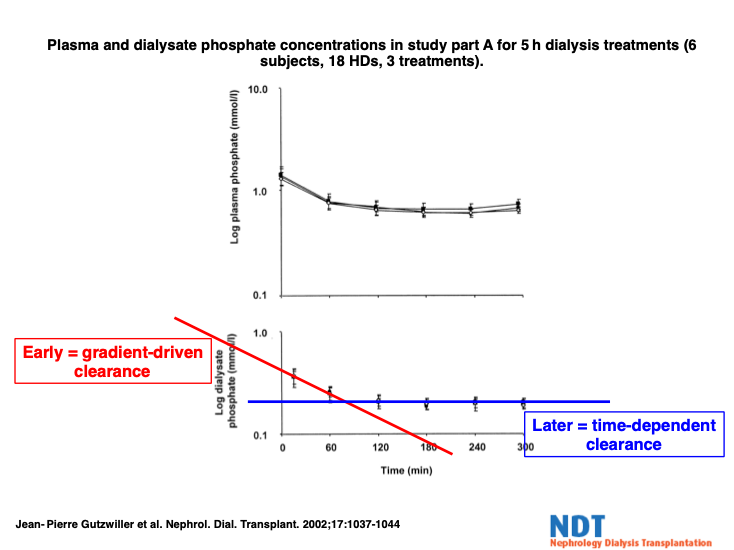
This suggests strongly, and is borne out in reality, that phosphate removal—as with other middle molecules—is governed by dialysis duration. The longer each dialysis treatment, the more efficient is the removal of phosphate and other similarly-duration-dependent middle molecules.
So, what the Gutzswiller study means is that longer is better.
Next: Think Volume
Here comes the now-familiar thread of several of my previous posts at this blog-site: volume. I have often previously blogged about volume in an effort to raise awareness that volume control is at least the equal of—or, in my view, is an even greater immediate threat to—the health and survival of dialysis patients than is solute control. I have often spoken of McIntyre’s seminal work in this area – especially of the impact of the rate of volume change through ultrafiltration while on dialysis. Rather than reiterate this again in detail here, can I refer you back to some of my previous volume blogs:
And … perhaps most of all …
What these all show are that—just as for solute removal—both duration and frequency matter…again! Thus … what McIntyre’s studies mean is that both more AND longer are better. I hope, by now, the penny has dropped!
I hope that this blog (among a wealth of research and evidence along these same lines) will begin to make providers think more carefully about the shortcomings of short and/or infrequent dialysis, and to better appreciate the failure of current practice to provide a cumulatively appropriate block of therapy per week. Short dialysis, infrequent dialysis, and worst, the combination of short and infrequent dialysis is simply selling our patients short.
Current 3 x weekly dialysis to a Kt/Vurea of 1.3 simply holds patients in CKD5. It holds them at the level at which they entered the dialysis pathway. It is a CKD5-equivalent. It also simply sustains them at the same level of health they had when we told them they would need dialysis. It hasn't “delivered” anything. It hasn't improved their lot. It hasn't fulfilled the promise: it has simply perpetuated the problem.
We can do better. We know we can. We have the ability. We have the tools. We have the practices.
While many now seem able to accept a “false fact” as fact in politics, we in medicine should not stoop so low. We should resolve to stop telling our patients the false fact that “current dialysis is as good as it gets.” It isn’t.
There is better, and it is our responsibility—as providers—to provide it.
References
1. Casino FG, Lopez T. The equivalent renal urea clearance: a new parameter to assess dialysis dose. NDT 1996 Aug; 11(8): 1574-1581
2. Gutzwiller J-P, Schneditz D, Huber AR, Scindler C, Gutzwiller F, Zehnder CE. Estimating phosphate removal in haemodialysis: an additional tool to quantify dialysis dose. NDT 1992 17(6): 1037-1044


Comments
John Agar
Aug 18, 2017 2:40 AM
I can only wish ... though I am sure my wish will not be granted ... that this discussion, and the blog that produced it, were read by more than the few who will ever likely see it. It goes to the heart of so many of the ills and ailments of our current 'care' modules, the way(s) we research their efficacy, and the many self-perpetuating falsehoods that emerge from that 'research' to shore up the status quo.
For those who do care, I suggest you consider copying the URL for this blog and its attendant discussion, and refer to it with whomever, wherever, and whenever you can.
David Glosser
Feb 17, 2017 1:19 AM
John agar
Feb 17, 2017 11:33 PM
Thanks so much for your thoughts. I understand exactly what you are concerned about ... namely, that US patients wouldn't accept more time on dialysis.
That attitudinal impasse is rooted in a range of mis-steps taken in US. These go, something like this ...
The first mis-step arose from the decade by decade contraction of dialysis time that occurred through a fatal misconception ... and, sadly, thousands upon thousands x fatal ... that Kt/V was all that mattered.
This was because of the erroneous belief that, mathematically, the same Kt/V could be achieved on the numerator line by shortening 't' and increasing 'K'. While this mathematically correct, this ignored that we are not mathematics, we are physiology ... and our physiology includes not just solute, but volume.
Relatively speaking, changing solute quickly is easier - much easier - than changing volume quickly. That is NOT to say it is necessarily 'good' to alter solute concentrations rapidly - it isn't - but fast solute change is certainly tolerated better than rapid volume change.
Change volume too quickly, and circulatory equilibrium is lost. When circulatory equilibrium is lost, our blunt physiological measures (like the BP) go haywire, while our more subtle measures (like tissue perfusion and tissue oxygenation) suffer more silently, but are equally as deadly.
So, the push to accept and drive shorter dialysis times spectacularly failed to consider volume!
While on a macro-financial scale, increasing 'K' to achieve shorter hour dialysis permitted ever greater patient throughput (and, as a cynic might observe, greater profit), it also led - insidiously - to the greater but largely hidden (or glossed-over) symptomatic disaster of circulatory distress. Patients were told (or led to think) 'this is what dialysis is' ... 'sorry, but suck it up!'
Because volume was being changed too rapidly, hypo-perfusion and hypo-oxygenation effects abounded. Patients cramped (peripheral hypo-perfusion) , went flat (macro-circulatory collapse), suffered cardiovascular events (myocardial hypo-perfusion) , felt nauseated (GI hypo-perfusion), and finished their short, brutal treatment mentally obtunded (cerebral hypo-perfusion).
In short ... that word again ... they felt terrible! And they died. Lots of them. Before their time ... that other word again. Their time was shortened ... and not just their dialysis time.
But, US clinics still sold (or were sold) the bigger 'K' and lesser 't' lie. And their patients learned to hate - and fear - dialysis. Indeed, there is a popular patient website that actually says so ... it's called 'I hate dialysis'.
Patients felt so bad that they craved, wept, argued, and demanded to 'come off' ... early. This led to even shorter, less efficient, more brutal, and less compassionate dialysis. It fed the lie.
Not so elsewhere. US dialysis times are not just shorter, but far shorter, than elsewhere. In my own neck of the woods, our mean treatment times are well over an hour per treatment longer - in centre. Ditto for Japan. Ditto for the UK, Germany, France, Canada. Meanwhile, logarithmically more patients are at home ... in Australia, the mean home patient pool is around 35% of all patients. In our unit, we have unbrokenly sustained >25% of all our HD at home since 2005.
Our patients know, from the outset, that their facility-based treatment time will be - on the average - between 4.5 and 5 hours ... it is part of the pre-dialysis education that begins at CKD4.
Better still, they are encouraged, where possible, to take dialysis home - where longer, slower treatments are possible in the sanctity of their own home. It is made clear that this is the BEST option. Finally, they are encouraged, where possible, to dialyse at home (we do not monitor) overnight, and at a minimum of second-nightly, as this offers the best dialysis of all. And our aggregate national survival is more than double yours, while our aggregate home survival is double that of centre survival.
Why? Because 'slow' dialysis recognises the importance of gentle volume change. It recognises that volume, not solute, rules dialysis speed.
This educational approach is called, for want of a better word, pre-conditioning!
But, you (the US) are now in a quandary. To be blunt, you have dug your own grave. Now that short, brutal, killing dialysis is so deeply ingrained in US dialysis culture, you have a huge struggle on your hands to alter (or correct) (mis)perception and (mis)expectation.
If clinic A is offering 3 hour dialysis treatments while clinic B round the corner is saying that their option will take 4.5 hours/treatment ... patients - poorly educated, and poorly prepared - will always take the shorter option. You can't blame them. But, while being brutally dialysed and dying early, they will never know better or be told there is a better way.
How can you change these erroneous practices? I wish that that were easy. It isn't. But it doesn't mean you shouldn't try ... bit by bit. By better pre-dialysis explanation. By teaching patients the difference between solute and volume. By explaining, patiently, why fast dialysis begets symptomatic dialysis and yields crappy outcomes.
Mel Hodge (a long-time carer for his wife Jane) wrote a very thoughtful opinion recently in AJKD ... suggesting that is dialysis were to be reimbursed by the hour, US culture would change rapidly. It is worth reading! Well worth it.
So ... hard to change? Yes! But ... imperative to change? Absolutely.
David ... be the first clinic to let your patients into the simple secrets of good dialysis. All I can say is that someone has to start somewhere.
David Glosser
Feb 18, 2017 3:59 PM
Thank you so much for your thoughtful reply. It was a very insightful read to say the least. I regret to say that you are right on. Any time that a patient of mine demands to come off treatment early- which is very frequent - I try to educate them as best I can so that they have the resources to make the best decision for themselves. And make no mistake, in the US, patients are the center of care, and if they don't want to do something, we cannot force them. So basically, the need for better education is absolutely essential, like you said. Thank you so much!
Henning Sondergaard
Feb 22, 2017 4:22 AM
I hate to burst your bubble.
But as a patient with more than 50 years experience in chronic illness as well as a thorough background as a healthcare professional (HCP) focusing on the mental and cognitive aspects of illness - and as a very active home HD patient, splitting my time between Europe and the US - I do believe my words have some weight when I say: Patients are NOT at the center of care in the US or anywhere else in the World. And sadly dialysis might just be one of the areas with most room for improvement - especially in-center dialysis which is a classic lesson in the psychological concept of learned helplessness. Which in this case works perfectly as a pointer for the opposite of patient centered care (PCC).
As you read on, David, I want you to know that this is not an indictment on you or the work you do. From what I read, you strive to do the best you can to your patients but you must know that you work within a system that is comparable to 3rd World countries. The US is among the worst in the Western World when it comes to treatment of dialysis patients - sad but true - and PCC in your country also scrapes the bottom.
So why do I think that you are not practicing PCC? Well, let me give you the best example I know. If you have as much as one patient on a 3 times a week 4 hour treatment regime you are not performing PCC. Period. You are practicing $-centered care.
The 3x4 paradigm was invented in the late 1960s based on a cost-benefit analysis that went something like this: how do we most effectively treat the greatest amount of patients without them croaking in droves? And the answer was 3x4. I dare ask, how many modern treatment regimes rest on such a specious and antiquated foundation? Unfortunately dialysis is the stepchild of nephrology, it's a money maker for the industry and is forgotten by most doctors who'd rather concentrate on transplants.
Having tried it myself for a very brief period of time, I can truly say that 3x4 amounts to torture. It is known among many patients as either 'shotgun dialysis' or - more morbidly - as 'death dialysis.' But trying to change the regime to a longer one without thorough CKD education geared towards the individual patient's level of understanding is even more torturous in my view. And trying to urge or coax patients to stay on the machine doesn't help anyone either. We're in a very delicate catch-22 situation. Damned if you do, damned if you don't. And I don't envy those of you HCPs who are in the thick of this. The only way I see out of it is education, education, education - both when it comes to patients and staff. But most of all, send patients home where they belong. Without opening up a whole new discussion, I want to say that in-center should always be the last resort when all other avenues (including conservative - or palliative - care) have been exhausted. Home first and in-center as the fallback option.
Yes, 3x4 is practiced in many parts of the world and not everywhere money is the main driver. In my country of Denmark as well as many other places I see a large measure of staff pity as the driver. Not compassion but pity. If they were compassionate they would not have started their first patient on this regime but given them some form of adequate dialysis, bringing them to CKD 3 or 4 on Dr. Agar's scale above. But pity is a useless emotion. For decades we have had a slogan in the disability world: "Piss on Pity" in other words, pity gets us nowhere - in fact, pity only keeps us suppressed and second rate citizens. Pity is based on fear, in this case, the fear of 'this could happen to me.'
On a more general scale PCC cannot happen the way we run the modern healthcare system. I dare say there was more PCC when I was a child in the 60s and 70, regardless of the far more paternalistic and Victorian approach to healthcare back then. What has gone wrong in the intervening years is the fact that healthcare has gone from 'care for the patient' to 'care for the disease.' There is no longer a relationship between the patient and the HCP. The relationship that does exist is between the HCP and the disease and it is measured in tests, trials, scans and lab results. The patient has become the (sometimes annoyingly meddlesome) host of said disease which is truly the item at the center of care.
This is in large part a side effect of the silo'ed approach to healthcare where all specialties have become more and more isolated from one another. Each one of them requiring an ever more myopic focus from doctors and nurses who are trained to forget about the whole patient - and especially the psycho-social aspects of the patient's life so they can concentrate on the purely biological aspects of the disease at hand. This is great for finding and curing some particular aspect of the patient but it is really, really bad for the overall health of the patient - and especially for those of us with complex chronic illnesses.
Like I said in the beginning, this is not a personal indictment. We are facing a huge cultural problem if we truly want PCC. It's like turning an aircraft carrier 180 degrees. It takes time, patience and a concerted effort. But I do think we all need to realize we are part of the problem. And we need to admit to ourselves what we are doing wrong. We also owe the patients to admit how we work within a system that is fundamentally flawed but we are trying to do our best. And if they want to help themselves live better and/or longer lives they might have to make some sacrifices in the short run but they will reap the rewards down the road if they change their schedule to something that is better for their bodies.
There is no doubt those of us doing home dialysis feel much better than those in-center. And I dare say every nation on Earth can expand their home programs, even New Zealand where HHD is 15 times as prevalent as it is in the US.
Dori Schatell
Feb 22, 2017 2:39 AM
The right to refuse any treatment, aspect of treatment, or participation in research is one of a tiny handful of absolute rights granted to US dialysis patients by Medicare. However, much as I WISH patients were at the center of care here, I fear that we are just starting to scratch the surface of shared decision making and true patient-centered care. At the moment, I think it's closer to corporation-centered care, but there are glimmers of hope on the horizon, such as the ESCO pilots...
David Glosser
Feb 22, 2017 4:06 PM
Your experience as a patient is the most valuable when it comes to knowing what exactly needs to change. You have knowledge and experience that I do not have. Thank you for understanding that I and many others try to do our best in a system that is hurting. I will continue to teach, teach and teach my patients. If I may ask, however, do you have a caregiver or someone to help you with your home hemodialysis? The reason I ask is because not having an assistant and/or fear is the most common reason that my suggestion for patients to consider home hemodialysis gets shut down. Many patients do not have a family member that is willing and ready to help provide care. What would you say to those patients if you were in my shoes? Thank you so much.
Henning Sondergaard
Feb 22, 2017 10:33 PM
I will gladly share my view on the carepartner (I like that better than caregiver which puts the person dialyzing in a passive role) requirement. I have been a paraplegic my whole life, until 15 years ago I took care of everything myself, I had no help with daily activities. Then I got an aide, then another and now I have what amounts to two hours help every day. When I started dialysis I asked to have my aide hours expanded because I needed help with my treatments. That was denied on the grounds that if you were doing HHD, you ought to be able to do it unaided.
Now, if a carepartner is a requirement in one country and it's deemed redundant in another, chances are both countries are equally wrong. It is not necessary but it might be nice. And that is my stance. I know of many people in the US who dialyze w/o a carepartner (off script?) so I know it can be done in certain clinics. My point is it should never stand in the way of giving people the best treatment possible - or the treatment that they wish to have.
To tackle the other issue, fear, I think that proper training and education can alleviate a lot of it. I know one thing that works really well, that is peer-to-peer interaction/education. There is nothing like talking to or seeing someone your equal doing things with ease you fear or don't understand. But there are many other ways of helping people alleviate their fears, many dependent on the specific situation.
I hope that eases your quandary. You are always welcome to ask more questions or leave comments - it's the only way we all learn from each other.
John agar
Feb 22, 2017 8:30 PM
The concept of a 'care-giver' is another very US-system one. We do NOT train care-givers. Other family members, friends etc. may occasionally be involved in some aspects of the dialysis of our home patients, but it is uncommon, here, for any third person ( or should that be second) to take a controlling role in delivering home dialysis ... we train the patient, and only the patient. We do not require a 'buddy', either to do or even to be present, and anywhere between 1/4 and 1/2 of our patients are single and self-dialyse alone. The effect of this is to self-empower. It imparts self-reliance ... pride even (in the good sense of the word. Pride that - yes - I actually CAN be the master of my own destiny. So, the concept that patients can't go home unless someone ELSE takes responsibility is just plain wrong. It is wrong on many fronts. Having a 'supporter' is great, and many significant others fill that void, where possible ... but a 'carer'? ... no.
David Glosser
Feb 23, 2017 8:02 PM
Thank you for the input you provided. I would definitely agree with you that it is good for patient's to provide self-dialysis at home even if someone else isn't present to help. If I were a dialysis patient, I would certainly rather HD at home over in-center (probably right before bed, I might add). Perhaps I should have used the word "care partner" instead of caregiver (I apologize), because I know that home HD patients in the US do a majority, if not most, of the treatment care themselves. My interactions with patients who do their hemodialysis at home is rather limited (probably 2 to 3 patients at the most), so I unfortunately cannot tell you with absolute certainty exactly how much of the treatment is done by the patient at home in the US. I want to make it clear, too, that I only speak for myself, not the whole of United States dialysis. I know that there are things that aren't being done in the US, but that doesn't mean that there aren't important people/ leaders in the US dialysis industry trying to change that. Even though in-center hemodialysis is much more prevalent in the US than in other countries, I'm confident that this will change. I have heard that FMCNA (the largest dialysis provider in the country) is pushing for more home hemodialysis. I've also heard that there is a push in the US to modify the reimbursement of hemodialysis so as to get more patients to dialyze in the home setting (reimbursement is huge). You can pretty much blame the whole thing on reimbursement. Anyway, I'm very glad I came across this post and got to read and converse with you all. It has been most educational and informative. I like knowing how other countries are doing things.
Jenny Flythe
Feb 07, 2017 8:01 PM
I always enjoy your commentaries and am certainly a proponent of longer treatments and slower fluid removal. The argument you lay-out is convincing, but critics would (appropriately) remind the audience that trial data is mixed. In that regard, could you provide some commentary and opinion on the recent JASN study evaluating longer dialysis and quality of life, among other outcomes? (Jardine, et al. J Am Soc Nephrol. 2017 Feb.
A Trial of Extending Hemodialysis Hours and Quality of Life. )
John Agar
Feb 08, 2017 9:07 PM
Much as I admire the effort good friends made to run this trial, I elected not to enter our unit patients - and I was not alone in this. As a non-believer in dialysis-modality RCTs but a believer in the many and ample observational studies and registry analyses around extended hour or frequency (high intensity) dialysis, I couldn't bring myself to ask of any patient where either I, the patient, or both believed that benefit would accrue from a long and frequent regimen (we encourage a 4-5/week x 8-9hr/Rx overnight home dialysis program) to then be randomised into a lesser hours/week option .. an often long-break-sustained treatment schedule with a total membrane contact time of <18 hrs a week.
As a result, and after much soul-searching … the trial was run by good friends, tried and true … our service was one of several who refrained from the trial – citing as justification our belief in more is better.
Before going to the trial itself, I want to make some observations about dialysis trials. In 2014, I was invited by the editor to submit a debate paper to the ASN monthly news and views publication ‘Kidney News’ supporting the 'no' case for the question ... “Are more randomised control trials needed to confirm the outcomes of home hemodialysis?”
The full text of my argument is available at ...
http://www.kidneynews.org/kidney-news/special-sections/home-dialysis/home-hemodialysis-do-we-need-more-randomized-controlled-data
In brief, the crux of my argument was as follows: while trials certainly play a vital role in evaluating the outcomes of a medical intervention like the comparison in outcome between a drug and a placebo, or between two drugs, head to head, and usually produce interpretable and reliable results ... trials that compare 'lifestyle' choice are, in effect, impossible.
Even if the dialysis prescriptions are the same, it is impossible to 'correct for' the myriad individual patient preferences that emerge and confound. Patients may either like - or dislike - the rosters, the staff or other patient inter-relationships (good and/or bad), the inflexibilities, travel issues (like cost, distance, parking, or time), the waiting times, the whole 'being cared-for' mentality, and the production-line feel of centre-based dialysis. Trying, then, to then compare these on any form of equal footing with the self-care freedoms – and anxieties, the travel and personality-clash free convenience of, but, for some, the training time, the ‘what if’ isolation and anxiety that may from time to time assail those on home-based care … all in a highly selected subset of HD patients that rarely (if ever) look like the ‘whole’ in either group, who then are enrolled randomly into a group they later wish they were not in … and finally, to extrapolate the always questionable findings to dialysis outcome across the full spectrum … I am sorry, but it doesn't wash with me. Well-intentioned, maybe … but interpretable, never.
No trial format will be able to balance for (or … in the language of the trialista’s … correct for) these incomparables.
In my view, it is actually fatuous to try.
But then … to add to ALL these variable variables the further multiple variables that form the differing regimes of duration, frequency and dialysis settings ... and any thought of a valid, interpretable, useful trial flies out the window.
This is the crux of why I think:
(1) RCTs in this domain will never clear the air but simply further muddy already opaque waters
(2) We should leave trials out of it, and simply ask any participant who has experience of each option what that experience has been
(3) Warts and all registry outcomes still have a lot to say.
As for the trial itself, I will add, here, some observations made by Eric Weinhandl, a epidemiologist from Minneapolis who worked with Allan Collins at USRDS for a number of years … his take on Meg’s ACTIVE trial publication includes … and I agree ...
1. The press release headline is really crude, though admittedly, the press release will be forgotten soon enough.
2. During follow-up, HD for at least 4x/week was about 40% in extended duration and 20% in regular duration, so this is more so a study about extending thrice-weekly session length; a lot of people in this study still have 2-day gaps.
3. EQ5D is not my favorite instrument, and beyond that, it is interesting that baseline scores on EQ5D were rather high.
4. Around 65% of patients were from China; it is not known how well-validated EQ5D is in Chinese dialysis patients, nor how much QOL might be modified, in light of some external factors [eg, air pollution].
5. The was a big improvement in physical component score (more hours vs. short(er) hours) from their SF-36 scores, but that's not even in the abstract.
6. p-values aside, LV mass index fell almost 10% with extended duration, and that's not really a "negative outcome".
7. One of the more vivid demonstrations of improvement in anemia management with intensive HD was the increase in Hb, yet with a corresponding decrease in ESA dose.
8. The usual (positive) changes in K, PO4, and oral med use were observed.
In addition, it is all getting very murky out there. If one reads a further (and rather late) assessment that came out last month in KI from the FHN2 trial group - a trial I still feel was fatally flawed - it reaches a different conclusion ... and this time in favour of extended hour home dialysis ... see ...
https://www.ncbi.nlm.nih.gov/pubmed/28094031
To be honest, you can interpret all this stuff whatever way you like and reach different conclusions! I critiqued the FHN2 some time ago in a blog at this site ... see ...
http://homedialysis.org/news-and-research/blog/97-a-sad-direction-in-home-dialysis-research-the-fhn-2-nocturnal-survival-analysis
Eric Weinhandl did likewise ... see ...
http://homedialysis.org/news-and-research/blog/137-what-can-we-em-really-em-conclude-about-the-fhn-trials-and-mortality-on-frequent-hemodialysis
My own final ‘observations’ include that I agree with Eric in all of these points. In addition, I still believe, as I published in Kidney News in September 2014 (reference above) that RCTs in complex, many-faceted, lifestyle-changing treatments like home HD are so riddled with biases and perversions of fact - recognised and/or hidden - that RCTs in lifestyle choices are likely MORE than LESS unreliable than registry studies.
Not only are RCTs in this domain unreliable, but I would contend that they should not be used to support any conclusions about which is better, on or the other.
Until such time that a better trial tool is devised - and RCTs re NOT that tool - then I remain a warts and all registry data supporter as, for all the faults of registries, they are a true view of everyone.
For this reason, I far prefer to believe in a paper for which I was a joint author - first author = Mark Marshall from Auckland, New Zealand … a paper that can be found at …
https://www.ncbi.nlm.nih/pubmed/26561355
Jenny Flythe
Feb 10, 2017 9:57 PM
Henning Sondergaard
Feb 22, 2017 11:28 PM
I agree with John in his view on RCTs. They are worthless in the situations we are talking about here. In fact, they are worse than worthless because many doctors (and sometimes also researchers) put way too much stock in them because they were taught so in school and because they never learned to think for themselves. And that poses a threat to their patients when they make the wrong decision based on poor research.
But that is not what I want to talk about. John mentions how EQ5D is not his favorite instrument. He proved to me earlier today how humble he is (but that's a different story) I think his humility got the better of him here. EQ5D is s*** In fact, I will say EQ5D is what s*** excretes, or the thing that lives on what it excretes. I am no fan of HRQOL questionnaires in general. I think they all try to quantify the unquantifiable. Just like RCTs don't work when measuring dialysis adequacy for various reasons, you can't quantify QOL. Period! And EQ5D shows my point in spades.
It purports that 5 questions can cover a person's quality of life. That in and of itself is ludicrous. To top it off I am doomed at the very first question. To answer truthfully I would have to check the worst category "I am unable to walk about." But there is nowhere to explain that I haven't 'walked about' since I was 10! And the same b.s. goes on with the next three categories, self-care, usual activities, and pain/discomfort. I personally don't score low in those categories, but I have plenty of disabled and/or chronically ill friends who would score a big round zero in any or all of them while at the same time living an active life the quality of which far exceeds the average Joe. As far as the last category goes, it's nothing short of unethical. Anxiety and depression are official diagnoses for serious mental diseases (whether I agree with that or not) and nobody should ever be put in a situation where they are forced to self-diagnose as they are in category five.
Like virtually every other QOL questionnaire this one assumes that the person filling it out started out an Olympic athlete and now their health got in the way of the way they used to live their life - and it's a tragedy. They also assume a direct correlation between (perceived or real) Ill health and QOL - a concept that many studies have shown is utterly flawed. Unfortunately far too many doctors and other HCPs assume that disability and/or poor health categorically is bad and undesirable for the individual. In disability studies that is called the "disability as tragedy" view. A normative approach that is as wrong as it is demeaning. Those who care to actually study the psycho-social aspects of illness all know things are way more multi-faceted. Many people have gained great things from being born with or from having contracted what others deem horrific diseases. But those are not stories worth mentioning since humanity has a need to feel pity to feel superior.
Unfortunately each and every one of these questionnaires rest on the idea of "disease as tragedy." The worst thing is that this sort of thinking becomes a self-perpetuating feedback loop that teaches the diseased they are pathetic and pitiable. QOL questionnaires are tools for the narrow-minded professionals that ultimately helps them feel superior while widening the gap between themselves and those they are supposed to help. We need to take people serious - chronic illness or not - and actually talk to them instead of handing them an inane piece of paper that has no validity or reliability.
That said, I think it's pretty obvious why the Jardine, et all. article is a waste of grant money. Too bad otherwise intelligent people are not able to see thru an instrument that so obviously is The Emperor's New Clothes. I look at it and think to myself, when do we start using bloodletting again?
Mel Hodge
Feb 03, 2017 8:56 PM
If your comfortable with some math, look up the MDRD equation on line and plug in your serum creatinine, race, sex and age and it will give you an an estimated glomerular filtration rate (eGFR). Then. look up Chronic Kidney Disease Stages -- each stage is defined by a GFR range -- and knowing your eGFR, you'll see your stage -- and how far down you'll need to get your serum creatinine to arrive at Stage 3. If you need it, get some help with the math from your nephrologist or dialysis RN.
John points out that you can get there with dialysis 7 days/week. My experience, as the care giver for my wife Jane, was that nocturnal dialysis 6 nights/week with a Fresenius 2008K machine got her comfortably into Stage 3, but fell short with a NxStage System One (we alternated for years between the two machines at different locations). The difference was apparent from both how Jane felt and in her serum creatinine. I'm convinced this difference stemmed from the dialysate flow rate to blood flow rate ratio which was 1.5 with the Fresenius machine and 0.36 with the NxStage machine. We were limited by using dialysate bags with NxStage, but I believe that this difference would disappear if NxStage could bring the dialysate rate up to match the Fresenius rate of 1.5 or more (in-center machines are usually run at 2.0).
The hard truth is that other than transplant, the only way you will feel just as good as you did before your kidneys failed is through long, slow, frequent dialysis at home. It entails an undeniable burden. Only you -- and those close to you -- can decide if its worth it. Jane and I never regretted our choice...
John agar
Feb 03, 2017 11:21 PM
Ant de Villiers
Feb 03, 2017 4:56 AM
I'm a believer, - upgraded from 4hr 3x per week to 6 hr @ 3x per week (pump 200, flow rate 300, G16 needle). Labs are favourable (except calcium). The 'more frequent, longer, slower' mantra makes 100% sense, as does 8 hours overnight whilst in deep blessed slumber 6 times per week.
BUT there's the rub. Dialysis, no matter how slow and gentle, disrupts the inner being (whoever that might be) and sleep is fitful and intermittent (at best) for me. I hope persistence and practice may bring results, but as a life-long light sleeper I'm not too optimistic.
John agar
Feb 03, 2017 10:56 PM