Improving Dialysis Patient Care: MEI Comments on the ESRD Proposed Rule

On June 25, 2023, the Centers for Medicare & Medicaid Services published a proposed rule entitled “CY 2024 Changes to the End-Stage Renal Disease(ESRD) Prospective Payment System and Quality Incentive Program CMS-1782-P.” The rule proposed to update the amount Traditional Medicare pays for dialysis for patients with kidney failure and acute kidney injury. It described proposed changes to the Quality Incentive Program (QIP) and ESRD Treatment Choices (ETC) Model. And, the proposed rule discussed CMS’ commitment to equity in healthcare and its goal to examine how its programs and policies may be contributing to healthcare inequities.

We submitted our MEI comment as a patient education and advocacy organization whose mission is to empower people with chronic diseases to manage and improve their health. We limited our comments to key sections others might not address, first supporting two proposals related to the ESRD QIP: converting the Clinical Depression Screening and Follow-Up reporting measure to a clinical measure under the Care Coordination Measure, and adding a reporting measure for Social Drivers of Health. We recommended that when a patient screens positive for any social driver of health, the clinic should report whether there is a follow-up plan. Further, we suggested use of a standardized SDOH survey, such as the Accountable Health Communities Health-Related Social Needs Screening Tool.
Our comment strongly opposed CMS’ proposal to remove the ultrafiltration rate (UFR) reporting measure from the ESRD QIP, citing evidence to support keeping a UFR measure:
The importance of fluid management to patients health and wellbeing
The increased risk of death and ER visits following the 2-day gap that occurs weekly for patients receiving standard in-center HD treatments
The relationship between higher UFRs and increased risk of all cause and cardiovascular death, which you can see in the chart below from a 2011 paper by Flythe JE et al:
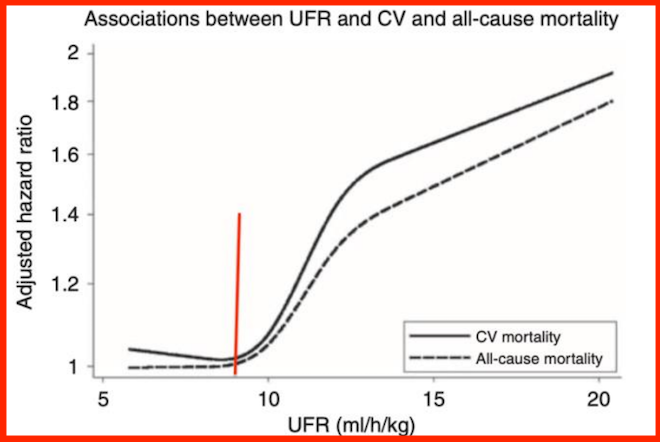
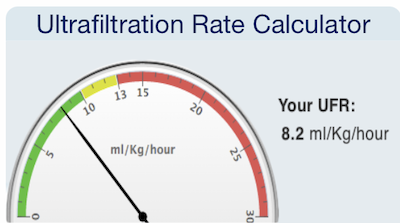
When we learned how strongly aggressive UFR predicted poor outcomes, we designed a free UFR calculator to help staff and patients determine a safe UFR for the amount of fluid to be removed during any given session. Patients who have large fluid weight gains need to do a longer or an extra treatment to stay in the green. This is much easier to do at home where scheduling home HD is more flexible. However, it is vital for dialysis clinics to avoid removing too much fluid too fast to protect patients from harm.
MEI’s comment included quotes like this one:
“I was about 15 minutes from getting off the machine, and the man next to me was having BP issues (low) had him in trendelenburg and had fluids going, then he got violently ill (throwing up) then started cramping real bad so a nurse helped him stand to try and work it out, not 5 seconds later he crumpled back in the chair and everyone is freaking out, I can’t listen to people throw up so I had my ear buds in but I felt the commotion on my chair as they were very close, so I turn around and he is back in trendelenburg and I can only see the whites of his eyes, next thing I know they were literally running the crash cart and privacy screen over, thankfully they were able to rouse him but barely, I left before the ambulance took him to the ER across the street, but that really scares me, in all my years of dialysis I’d never seen anything like that.”

MEI’s comment shared pertinent sections of the ESRD Conditions for Coverage (regulations) and the ESRD Interpretive Guidance, including the requirement for the dialysis team to assess and reassess patients’ fluid management and develop a plan for how to address identified issues. It also referred to the CMS Measures Assessment Tool that says to avoid UFR ≥ 13 mL/kg/hr. You can see why by looking at the chart above.
Our comment provided data to support continued collection of data on UFR. The United States Renal Data System showed hospitalization rates and ER visits that had been stable for some time declined in 2020, the first year dialysis clinics had to start reporting patients’ UFRs. We included references on such topics as:
UFR, Symptoms During Dialysis, Mortality, Dialysis Withdrawal
UFR (different levels) and Mortality
Sudden Death & Kt/V, Short Treatment Time, UFR
UFR & Dialysis Recovery Time
UFR & Decline in Residual Kidney Function
We closed out our comment on UFR with this recommendation:
“Rather than eliminate a measure for the critical metric of UFR, MEI strongly urges CMS to convert the UFR reporting measure to a clinical measure and require dialysis facilities to report data on in-center HD patients’ symptoms during and between dialysis treatments (e.g., low blood pressure, syncope, cognitive changes, headache, cramping, nausea, vomiting) and complications (e.g., loss of residual kidney function, clotted vascular access, emergency department visits, hospitalizations, and cardiovascular event or death) that occur following one or more dialysis sessions when UFR exceeded 10 mL/kg/hr.” We offered this rationale: “Collecting UFR AND symptom/problem data allows researchers to perform a national analysis of differences in outcomes associated with UFR >13 mL/kg/hr compared with UFR <10 mL/kg/hr. Failure to collect these data eliminates a major opportunity to improve hemodialysis care.”

Our final comment focused on our support for the Transitional Add-On Payment Adjustment for New and Innovative Equipment and Supplies (TPNIES) to include a device called the Buzzy® Pro. The FDA-approved Buzzy device has been used in dialysis to reduce needle fear and pain. We included patient quotes to illustrate needle fear and needle fatigue. Many clinics do not provide topical anesthetics (included in the Medicare bundle) or may not tell patients how to correctly use them. With Buzzy, timing is not an issue. A home HD patient can use a less costly, single-person version of the Buzzy. The Buzzy Pro was designed for multiple-use settings, and can be disinfected between patients. We concluded our comment with this statement: “We know we share the same goal as CMS of enabling patients with chronic diseases to live their best lives possible, and we believe the Buzzy Pro is a step in the right direction. TPNIES aside, we ask that CMS consider other ways to encourage research and early adoption of technologies like the Buzzy Pro, perhaps by listing it as an alternative to other topical anesthetics in the Bundle.”
The deadline for comments was August 25. When you visit the comment site, you can read any or all of the 346 comments. A search field allows you to look for comments by an individual or organization.


Comments