Vein Dilation Before Fistula Surgery: the PINCH and FACT

BACKGROUND
More than 100,000 Americans initiate hemodialysis (HD) each year, and more than 80% of patients use central venous catheters (CVC) as their initial vascular access, with a few using a surgical or endovascular arteriovenous fistula (AVF). The proportion of AVF use in new dialysis patients has remained below 20% over the past decade. Many reasons have been described for poor fistula success, including:
-
Lack of patient/physician education
-
Poor surgical results
-
Poor vein size in the arm, resulting in poor AVF development or more procedures.
Recently, there has been an international interest in making arm veins larger before AVF surgery. AVFs are universally acknowledged to be the safest way to access the bloodstream for hemodialysis. Successful AVF creation requires the preservation of native veins, which must dilate after surgery. This applies to both surgical and endovascular AVFs. Guidelines suggest that all suitable forearm and upper-arm veins should be preserved in patients with chronic kidney disease (CKD), avoiding both peripheral and subclavian venipuncture for any reason.

Despite this, construction of a viable AVF is often complicated or prevented by veins of insufficient diameter or quality. An AVF must ‘mature’ after creation: the vessel must dilate sufficiently to carry a high blood flow, and the walls of the outflow vein must thicken and become strong enough to endure repetitive needle cannulation and vein dilation to remain patent and accommodate repetitive needle cannulation. Vein dilation is one of the key requirements for AVF maturation, although blood flow rates, vessel depth, and the length of the accessible segment of vein are also important. Most surgeons require that the target vein for an AV fistula have a diameter of at least 2.5-3.0 mm in either the upper or lower arm. Unfortunately, many arm veins fail to dilate and enlarge sufficiently after AVF creation. When AVFs fail to mature, patients are subjected to additional surgery and invasive percutaneous procedures, resulting in hospital readmissions, delayed care, and increased costs.
Recently, the importance of vein size before AVF surgery has become an important topic globally. This has resulted in 2 very important trials to help understand the importance of vein dilation before AVF surgery. Hopefully, these trials will show that vein dilation before AVF with simple exercises or even a new novel device can help patients receive AVFs that develop and mature.
The PINCH Trial
Dorien Wilschut
Koen E.A. van der Bogt, MD
The Hague, The Netherlands
A popular approach over the years has been to encouragepatients to develop their veins by exercising their arms before and after AVF surgery, with the expectation that exercise will increase blood flow to the limb and that this will dilate veins. Forearm exercise has shown potential as stimulus for increasing blood vessel size in patientswith end-stage renal disease (ESRD) and may promote maturation of AVFs in the upper limb when applied postoperatively. Small diameter blood vessels and low blood flow are known reasons for non-maturation of AVFs. Forearm exercise has proven to acutely increase the diameter of the cephalic vein in dialysis patients with recently created AVFs. When applied routinely by ESRD patients without AVFs, forearm exercise sustainably improves cephalic vein volume two-fold within six weeks. Based on small, non-randomized studies, international guidelines recommend regular forearm exercise in the first months after AVF creation. This trial hypothesis is that when these exercises are applied pre-operatively, causing larger blood vessel size and increased blood flow, this may facilitate more distal AVF creation, and better maturation.
Methods:
The PINCH trial (refers to “pinching”) is an investigator-initiated, multicenter, single-blinded, randomized controlled trial with 1:1 randomization to perform supervised forearm exercises compared to no exercise 6 weeks pre-operatively before creation of an AVF. Forty patients receiving an AVF will be included. The main study endpoints are:
-
Blood vessel diameter (cephalic or basilic vein and radial and ulnar artery)
-
AVF surgical plan (radiocephalic or brachiobasilic/cephalic)
-
Three-month (assisted) maturation rate.
The burden of the performed forearm exercises will be evaluated using Kidney Disease Quality of Life (KDQOL-SF Dutch version1.2) and exercise specific questionnaires. The PINCH study is the first randomized trial to evaluate the effect of pre-operative, supervised forearm exercises on vein diameter and fistula maturation in hemodialysis patients.
Study Design and Treatment
 This study is designed as a multicenter randomized controlled study in subjects who are scheduled for creation of an AVF for maintenance hemodialysis, based on standard care criteria. As part of regular care, a pre-operative duplex study of the arteries and veins of both arms following a strict protocol will be performed. Following inclusion, 6-8 weeks prior to surgery, patients will receive the forearm training gear: hand grip trainers coupled to an application, using an e-device. Next, they will receive instruction and training schedule containing exercises for 30 minutes per day, in 3 sessions of 10 minutes, and 20 squeezes per minute, for 6 weeks.
This study is designed as a multicenter randomized controlled study in subjects who are scheduled for creation of an AVF for maintenance hemodialysis, based on standard care criteria. As part of regular care, a pre-operative duplex study of the arteries and veins of both arms following a strict protocol will be performed. Following inclusion, 6-8 weeks prior to surgery, patients will receive the forearm training gear: hand grip trainers coupled to an application, using an e-device. Next, they will receive instruction and training schedule containing exercises for 30 minutes per day, in 3 sessions of 10 minutes, and 20 squeezes per minute, for 6 weeks.
Patients will be referred to a physiotherapist for once-weekly group sessions where training will be monitored and intensified. Moreover, grip force will be objectified using a dynamometer and forearm circumference (5 cm distal from the elbow crease) will be measured. Questionnaires on kidney disease quality of life-short form (KDQOLSF) and burden of the exercises will also be performed.
After 6 weeks, the duplex investigation will be repeated and diameter of cephalic vein at pre-specified locations, diameter of radial artery and blood pressure at both sides will be measured. Then the patient will be contacted by telephone by the vascular surgeon who can finalize the operative plan. The operation will be conducted at 6-8 weeks following initiation of forearm training. Follow-up visits will be routinely planned at 6 weeks postoperatively accompanied by a duplex investigation. At 12 weeks, a follow-up telephone consultation by one of the vascular surgeons will be done to ask whether the AVF is being used for hemodialysis, and to check for non-surgical patency-promoting interventions, such as balloon angioplasty.
The FACT Trial
The Fist Assist Clinical Trial
Dr. Mary Hammes/ The University of Chicago (PI)
Dr. John Lucas/Greenwood, MS
Dr. Sanjay Desai/MS Ramaiah Medical College, Bangalore, India
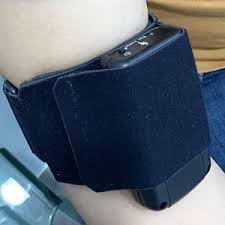 There is an urgent need to find more effective methods to promote vein dilation and develop cost effective, non-invasive devices to help mature fistulae clinically, and help AVF mature. Based on the basic science and clinical science of vein adaptation to compression, a novel new device has been created by vascular access surgeon, Dr. Tej Singh from Silicon Valley, CA. The Fist Assist® device (US Patent 8231558) is an external medical intermittent pneumatic compression device that can apply intermittent pressure to the arm. The FACT trial will evaluate the effects of using the Fist Assist® device to develop veins prior to AVF surgery. The device is presently marketed for sale in Europe, India, Australia, and New Zealand on Amazon.
There is an urgent need to find more effective methods to promote vein dilation and develop cost effective, non-invasive devices to help mature fistulae clinically, and help AVF mature. Based on the basic science and clinical science of vein adaptation to compression, a novel new device has been created by vascular access surgeon, Dr. Tej Singh from Silicon Valley, CA. The Fist Assist® device (US Patent 8231558) is an external medical intermittent pneumatic compression device that can apply intermittent pressure to the arm. The FACT trial will evaluate the effects of using the Fist Assist® device to develop veins prior to AVF surgery. The device is presently marketed for sale in Europe, India, Australia, and New Zealand on Amazon.
Fist Assist® is a self-contained, miniaturized, wearable intermittent pneumatic compression device. Patients are able to apply and remove the device themselves using only the contralateral hand. It is battery-operated and has a single control to turn it on or off. All pressure and timing parameters are preset at the factory. The bladder is inflated to a pressure of 60 mmHg and held for 20 seconds then deflated to 10 mm Hg pressure for 55 seconds before the next inflation.
This first proposal of presurgery vein dilation outlines important research that will help to determine the optimal role of the Fist Assist® device in improving vascular access outcomes in patients receiving HD, by investigating the effects of the device used before AVF surgery. Early use of this device could potentially allow for vein dilation in patients with advanced chronic kidney disease, preparing veins for AVF creation and improving primary AVF outcomes. Investigation with this device may uncover a safe and noninvasive way to improve outcomes for ESRD patients, and provide important insights into the biology of successful AVF maturation.
Study Design and Treatment
This study is designed as a multicenter randomized controlled trial in subjects who are scheduled for creation of an AVF for maintenance hemodialysis in the future, based on standard care criteria.
Hypotheses of FACT:
Use of Fist Assist® before AVF creation will enhance superficial vein dilation, resulting in increased absolute vein diameters at 3 months when compared to baseline values.
SPECIFIC AIM:
To determine whether the use of the Fist Assist® device for 360 hours over 90 days by patients with advanced CKD prior to AVF surgery results in significant increases in cephalic vein diameters prior to AVF surgery. Patients will have a preliminary evaluation of their cephalic vein, measured 5 cm proximal to the styloid process of the radius, and also at the antecubital fossa.
Intervention
The Fist Assist® device will be assigned to each enrolled subject by randomization code. All subjects will be asked to use their assigned device twice daily, for two 2-hour sessions, once in the morning and once in the evening. Patients will apply the device to the non-dominant arm above the level of the elbow, and to keep a written log to record use, complications and any problems. Patients will have follow up examinations with measurement of cephalic vein diameter at three months after enrollment.
Outcomes
The primary outcome will be the difference in vein diameter before and after Fist Assist®use. The goal is to see vein dilation over .4mm and to see how many veins approached the minimal size of 2.5 mm for fistula placement. Secondary outcomes include:
-
Whether or not an AVF was created (versus a synthetic graft or use of a central venous catheter)
-
Whether the resultant vascular access matured sufficiently to be used for HD.
-
Subsequent exposure of patients to central venous catheters during their first year of HD.
Conclusions:
To improve AVF placement and success for hemodialysis, presurgery vein enlargement is becoming a very interesting science. In the coming year, the Pinch and FACT trial will shed some important data on the benefits of vein dilation before AVF surgery and hopefully better AVF maturation and long-term results for patients with renal disease. Both trials will address vein dilation with their own approach: exercise vs pneumatic compression. Regardless of the approach, the vein dilation before AVF placement will be well received for both surgical or endovascular AVF creation.
|
Trial |
PINCH |
FACT |
|
Region |
Europe |
USA and India |
|
Type |
Multicenter,single-blinded, randomized |
Multicenter,single-blinded, randomized |
|
Enrollment |
20 and 20 controls |
45 |
|
Timeframe |
6 weeks before surgery |
3 months before surgery |
|
Method |
Supervised forearm exercises with hand grip trainer |
Fist Assist FA-1 Device in upper arm |
|
Daily program |
30 minutes per day (20 squeezes per min)/physiotherapist weekly |
Device 4 hours per day application |
|
Data Log |
Grp force, Kidney disease QOL and exercise buden |
Daily log of complications |
|
Study targets |
Arm veins and arteries |
Cephalic vein size and perforator vein |
|
Primary Endpoint |
Per Patient increase in cephalic or radial artery diameter |
Cephalic vein size greater than .4 mm |
|
Secondary Endpoints |
Cephalic vein diameter 6 weeks after surgery, Per patient change in cephalic or basilica vein diameter from surgery to 6 weeks, AVF flow 6 weeks after surgery |
Percentage of veins approaching 2.5 mm or 3.0 mm for AVF creation, successful surgery with AVF, matured fistula, and catheter contact time. |


Comments